
for use after liposuction, and the
only device that is FDA-cleared for
contracting subcutaneous tissue.


for use after liposuction, and the
only device that is FDA-cleared for
contracting subcutaneous tissue.


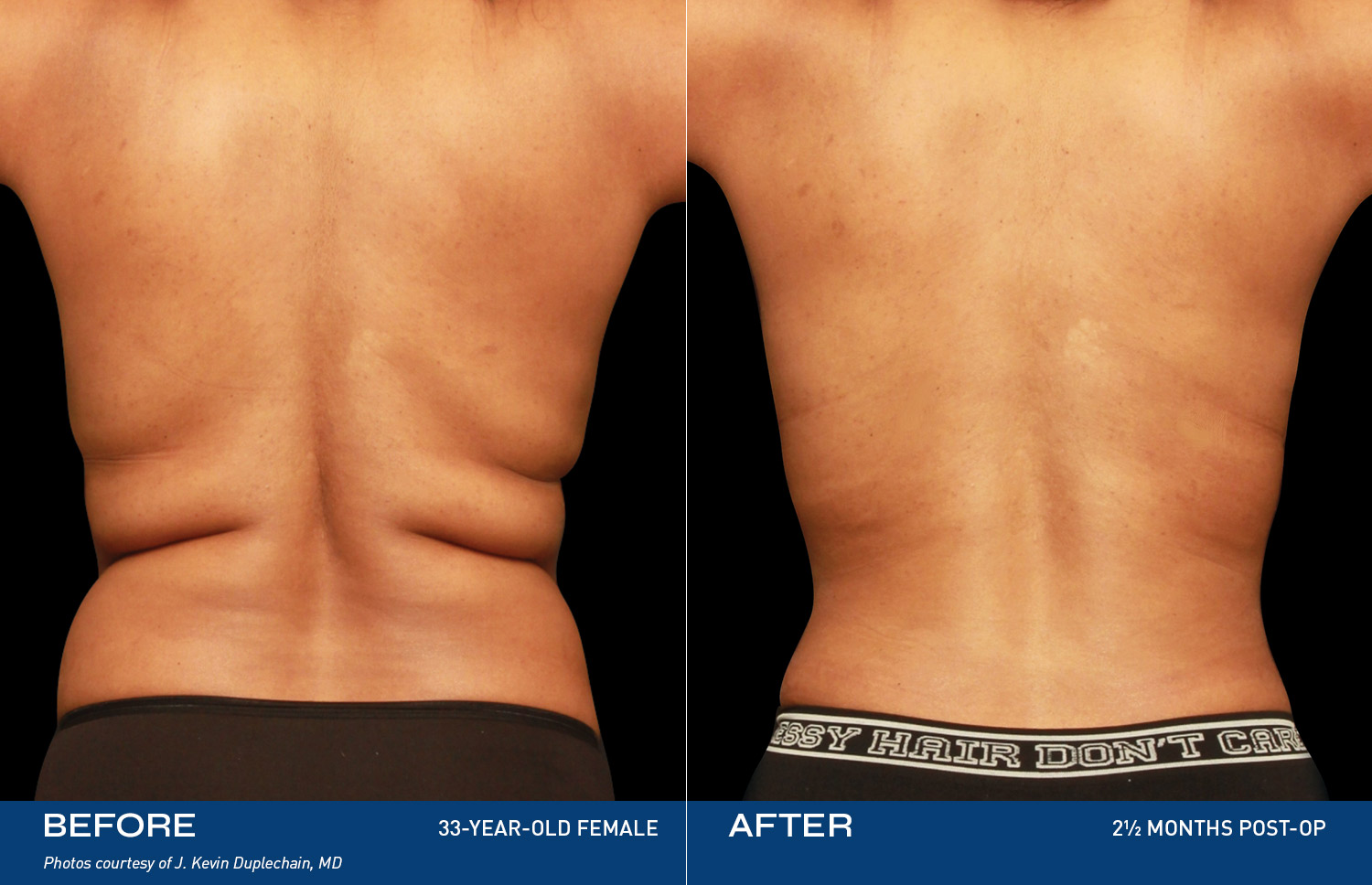
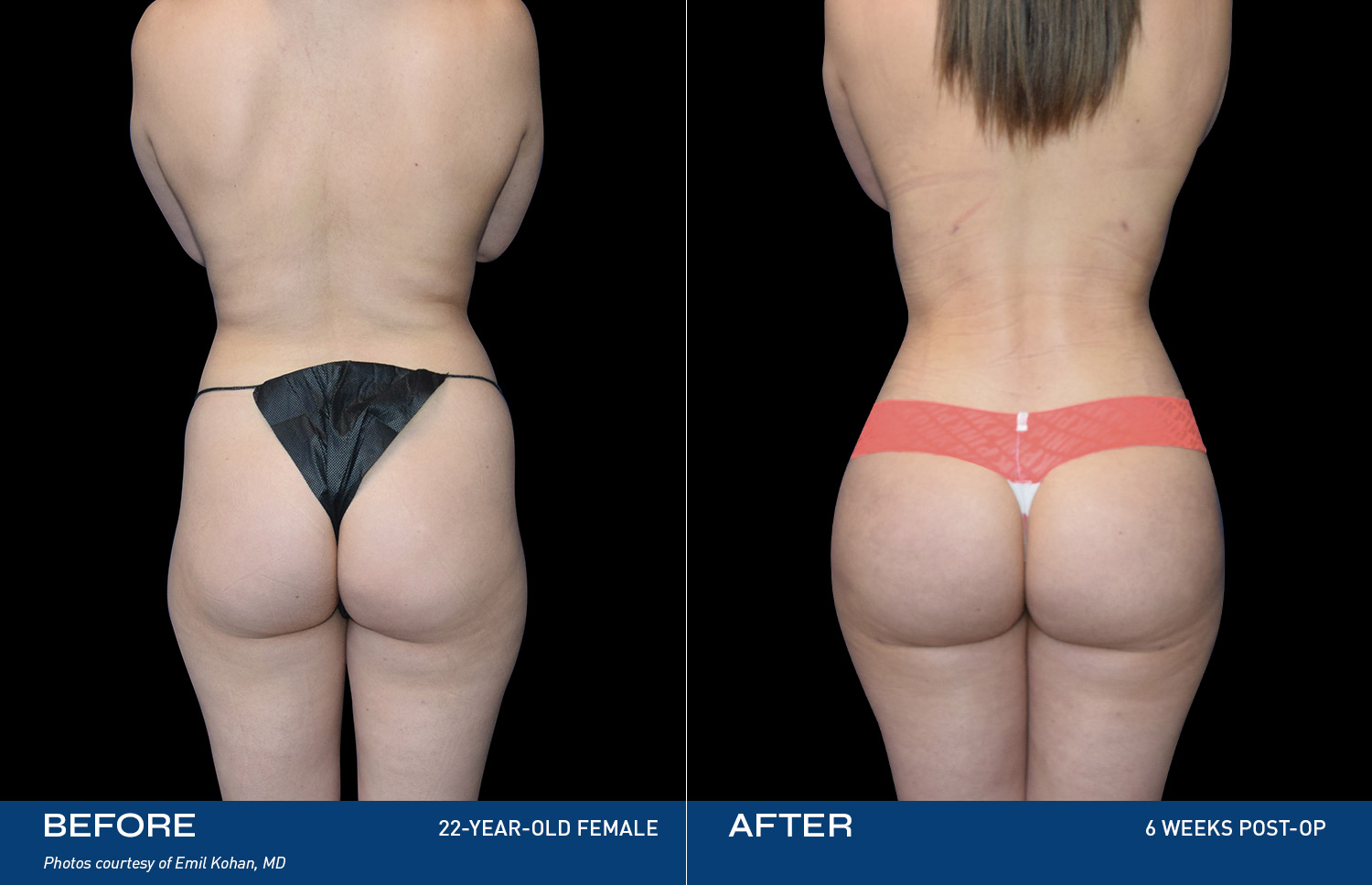
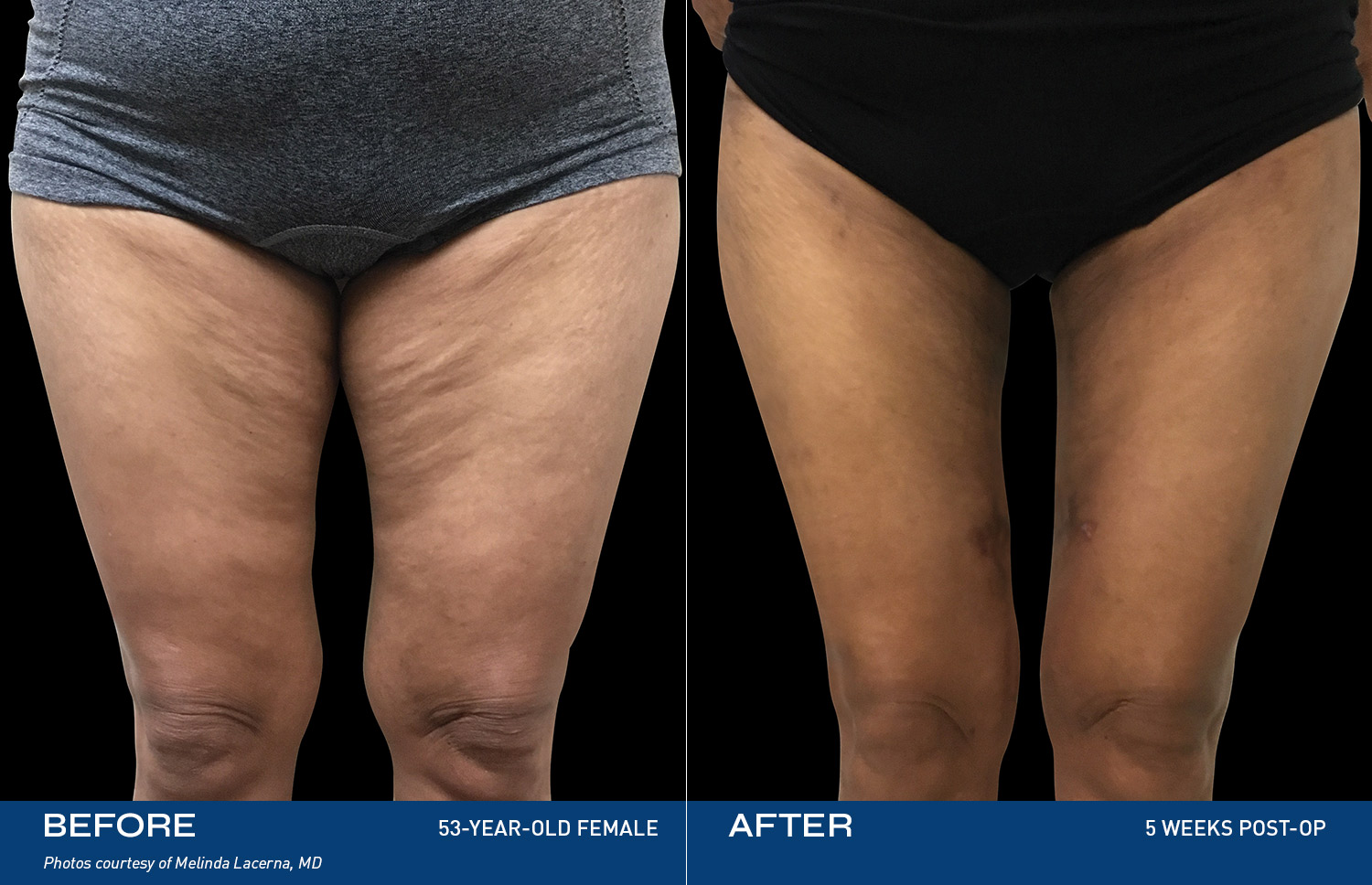
REAL RESULTS
PHYSICIAN FINDER
Enter your Name, Email and Zip Code below to find physicians near you that use Renuvion.

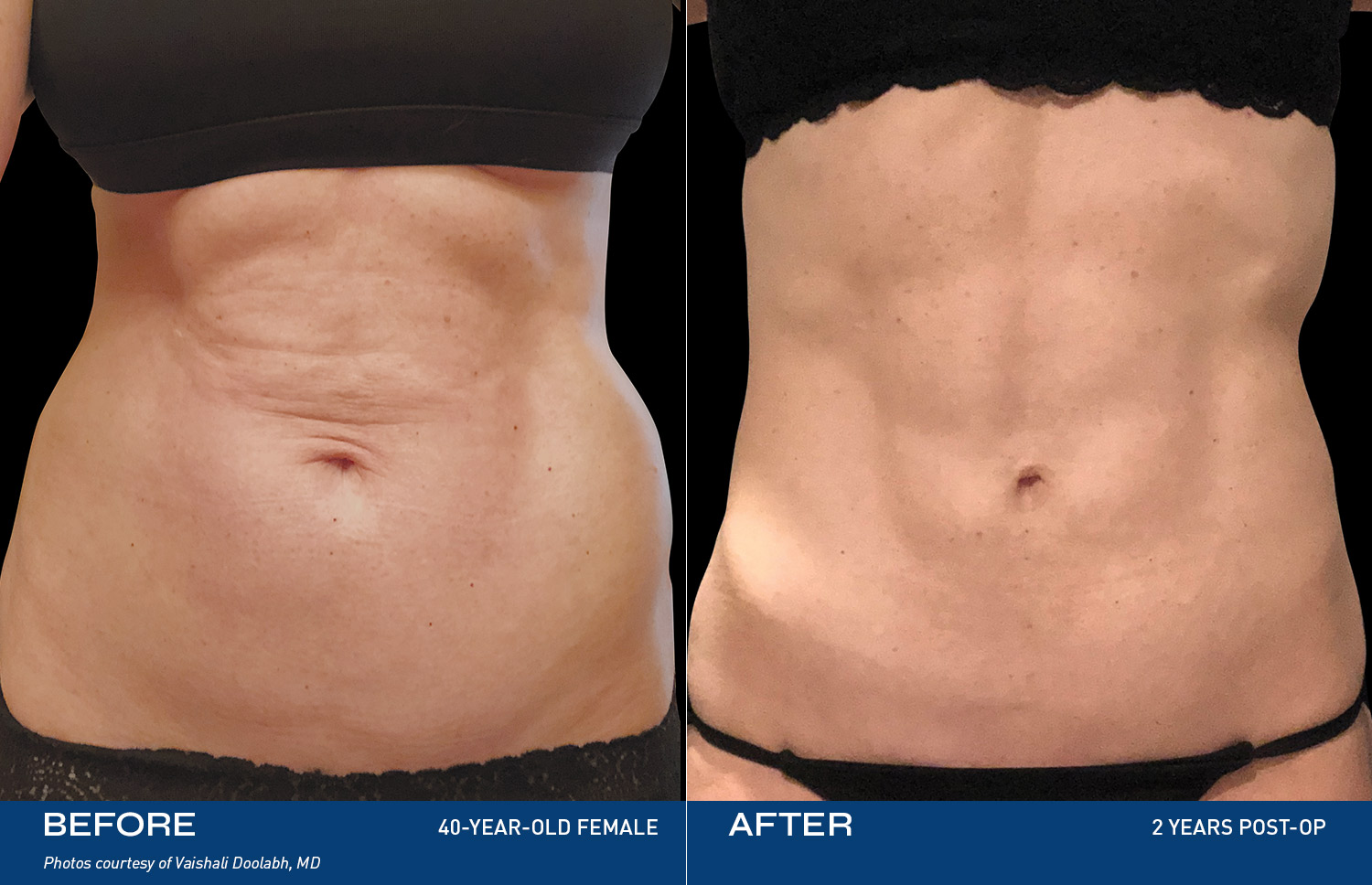
CONTOURING
The Renuvion Body
Renuvion is the only device that is FDA‑cleared for use after liposuction, and the only device FDA‑cleared for contracting subcutaneous soft tissue.
HOW IT WORKS
The wand is inserted under the skin and delivers a unique combination of radiofrequency energy and helium plasma to the collagen-rich tissue that’s a primary cause of loose skin. This energy contracts these collagen fibers and pulls the skin down, closer to the muscle, for a more contoured appearance. Renuvion is the result of decades of research, evidence-based medicine and clinical data.
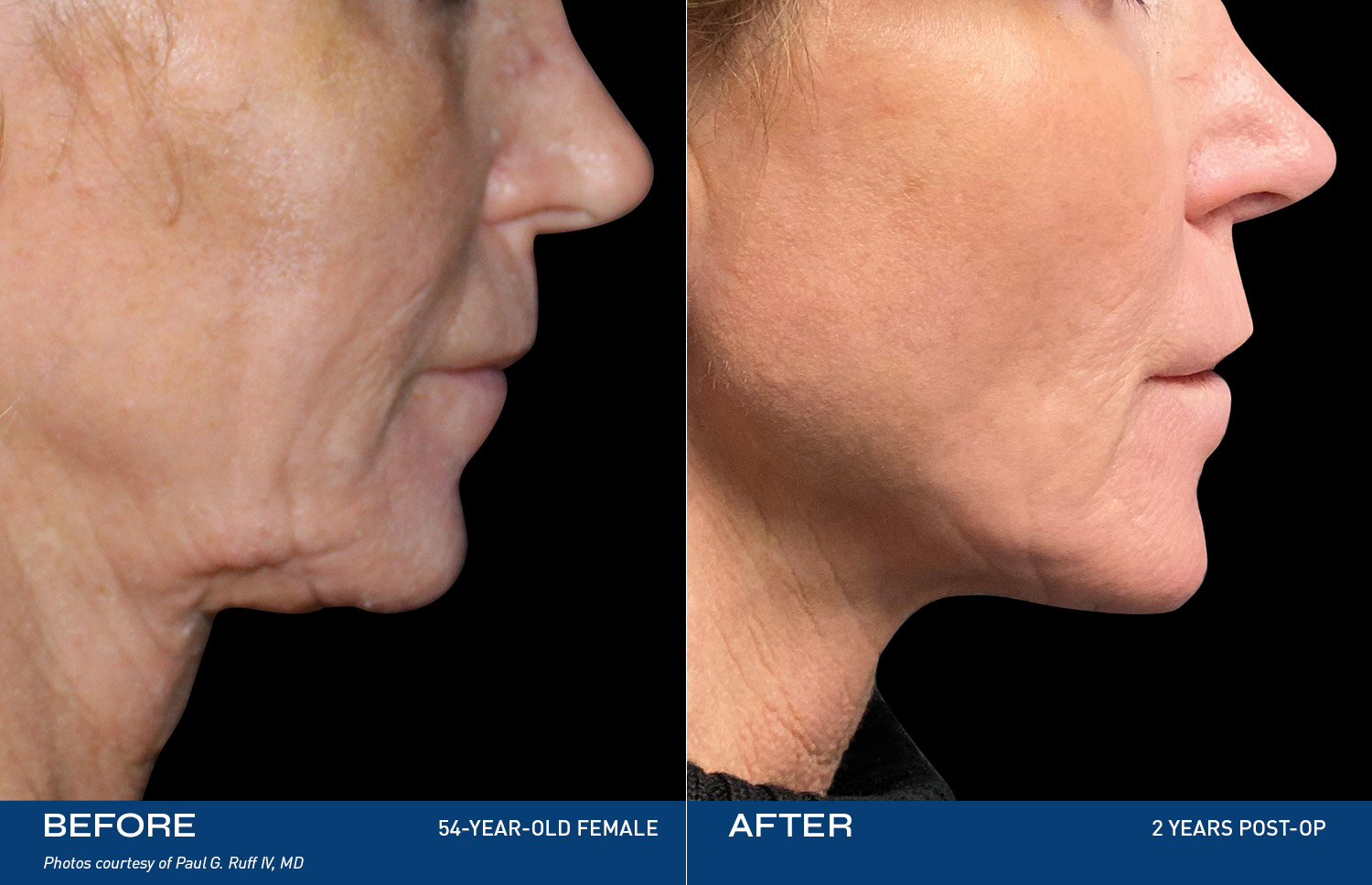
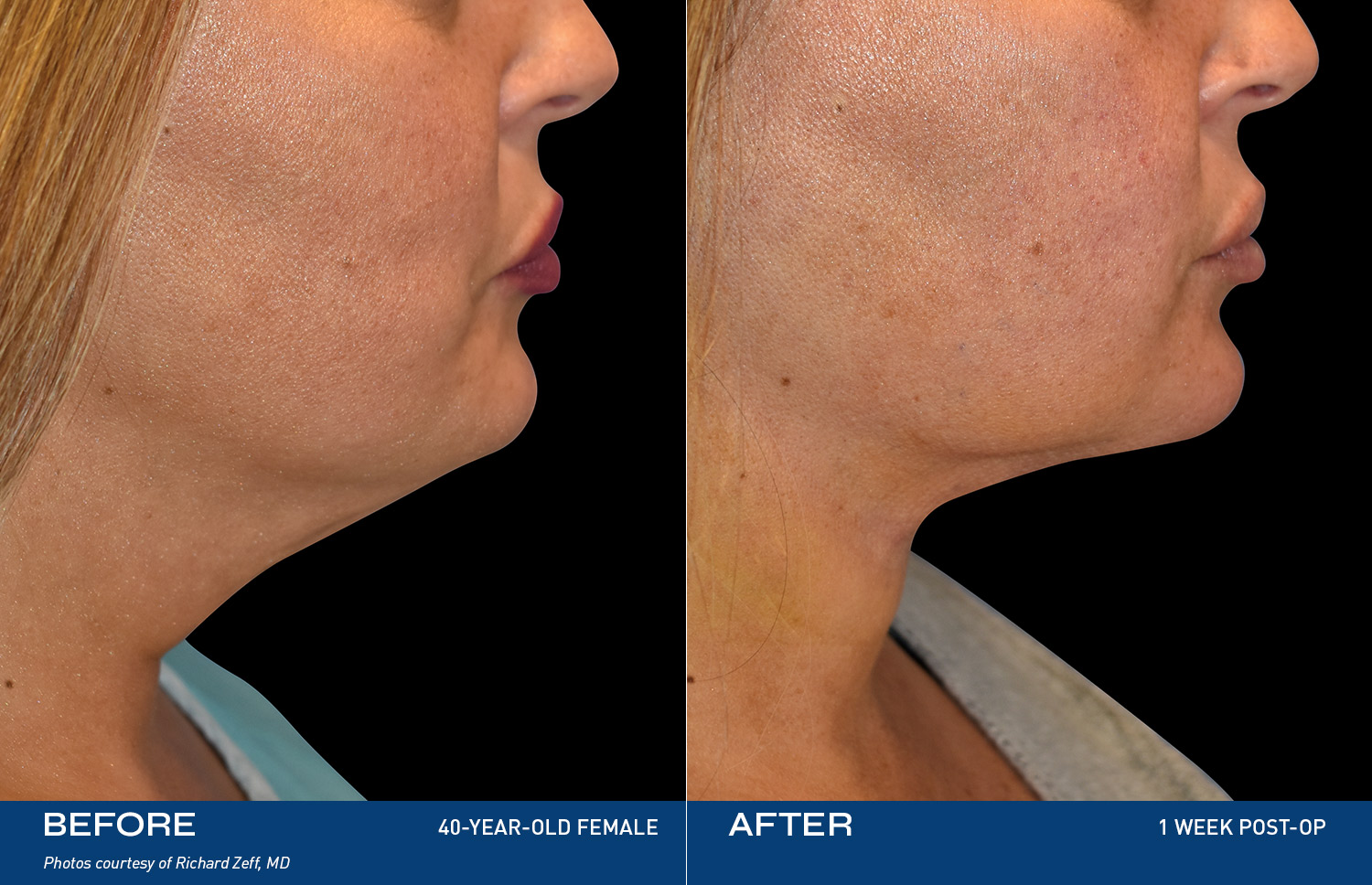
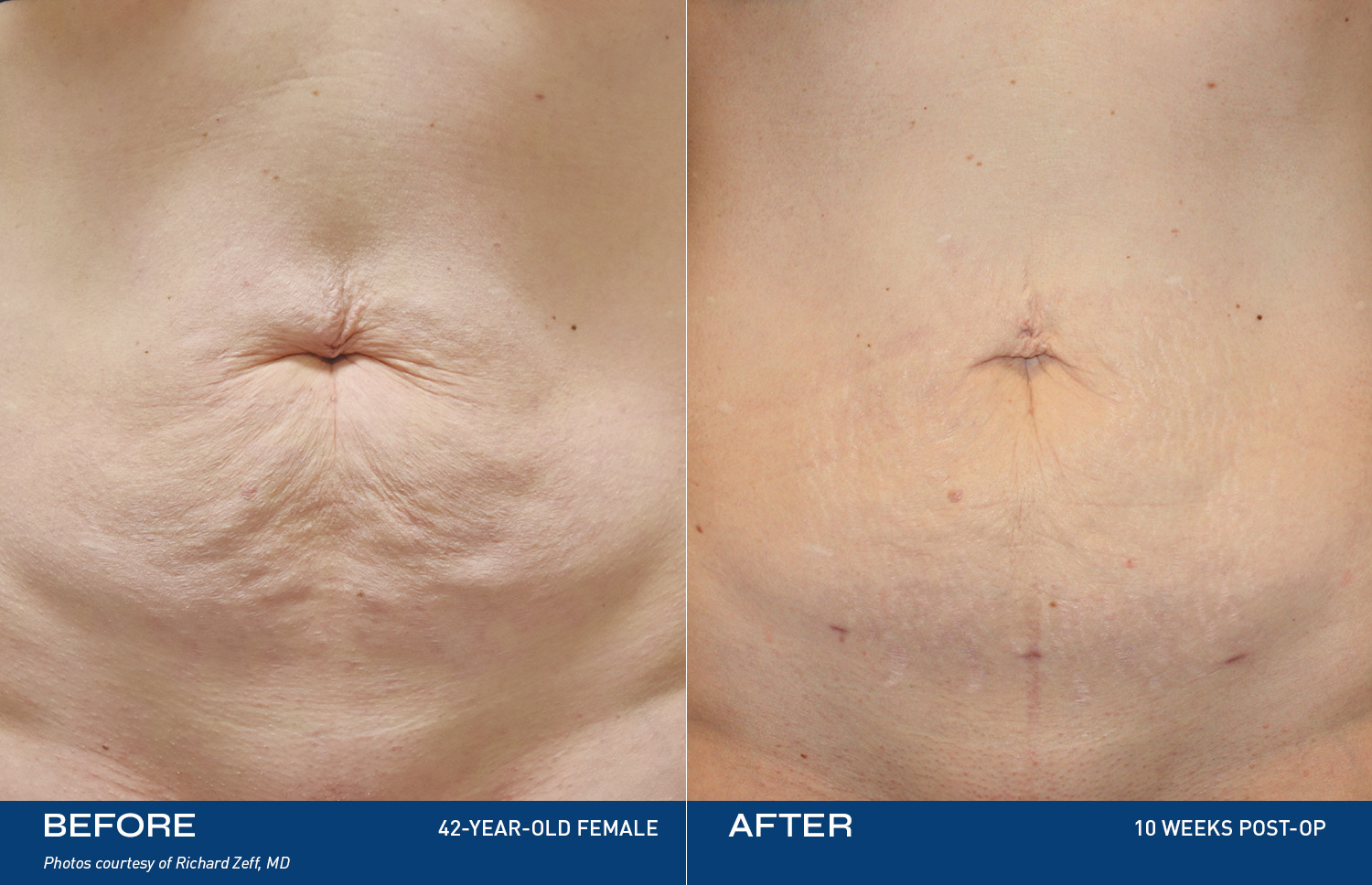
REAL RESULTS
BEFORE & AFTERS
"I tried so many things, but my skin was still loose. After Renuvion my neck and jawline look so smooth and it feels tight like it used to. I'm so happy with the result."
- Renuvion Patient
doctor
Testimonials
doctor
PAUL NASSIF
doctor
Tabasum Mir
doctor
Leif Rogers
doctor
PAUL RUFF
doctor
Alexis Delobaux
doctor
Gregory Buford
FREQUENTLY ASKED QUESTIONS
Renuvion is a single treatment. Results may be seen in as little as days or weeks and will continue to improve throughout the healing process.
Depending on the area of the body treated, the use of Renuvion only adds a few minutes per area to a standard liposuction case.
LATEST NEWS
AS SEEN IN











The Renuvion APR Handpiece is intended for the delivery of radiofrequency energy and/or helium plasma where coagulation/contraction of soft tissue is needed. Soft tissue includes subcutaneous tissue. The Renuvion APR Handpiece is intended for the coagulation of subcutaneous soft tissues following liposuction for aesthetic body contouring. The Renuvion APR Handpiece is indicated for use in subcutaneous dermatological and aesthetic procedures to improve the appearance of lax (loose) skin in the neck and submental region. The Renuvion APR Handpiece is intended for the delivery of radiofrequency energy and/or helium plasma for cutting, coagulation and ablation of soft tissue during open surgical procedures. The Renuvion APR Handpiece is intended to be used with compatible electrosurgical generators owned by Apyx® Medical. Not all indications are approved in all markets; check with your local sales representative for further information.
As with any aesthetic procedure, individual results may vary. As with all energy devices there are inherent risks associated with its use. Risk associated with the use of the Renuvion APR may include: helium embolism into the surgical site due to inadvertent introduction into the venous or arterial blood supply system, unintended burns (deep or superficial), pneumothorax, temporary or permanent nerve injury, ischemia, fibrosis, infection, pain, discomfort, gas buildup resulting in temporary and transient crepitus or pain, bleeding, hematoma, seroma, subcutaneous induration, pigmentation changes, increased healing time, scarring, asymmetry and/ or unacceptable cosmetic result. Please see the instructions for use for more detailed information.
References:
- FDA 510(k) Premarket Notifications K230272 & K223262.
- Duncan DI and Roman S. Helium Plasma Subdermal Tissue Contraction Method of Action. Biomed J Sci & Tech Res 31(2)-2020. BJSTR. MS.ID.005075.
